Ultherapy – Chicago & Skokie
Chicago Dental Arts is among the first in the Chicago area to offer the Ultherapy procedure for lifting and tightening facial and neck skin—without unnecessary surgery! We use tried-and-true ultrasound energy to strengthen your skin from the inside out, in a completely non-invasive way. We promise that you’ll love the results.
In less than an hour – and without any downtime – you’ll be well on your way to tighter, better-fitting facial skin. Ultherapy has been established as a safe and effective clinical treatment after more than 350,000 worldwide treatments. After just one treatment, with no downtime, your skin will gradually reflect a more lifted and toned appearance, reversing the affects of the body’s natural aging process.
What is Ultherapy?
Did you know that Ultherapy treatments are the only non invasive treatment cleared by the FDA to target the loose or sagging skin in your neck, chin and brow? Through a micro-focused ultrasound, the Ultherapy procedure lifts and tightens your loose skin, leaving you with a youthful and radiant appearance.
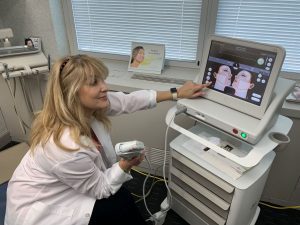
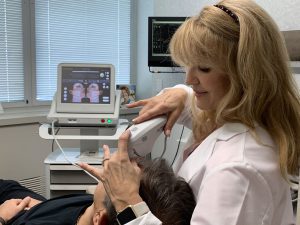
Ultherapy in Chicago & Skokie, IL
Contact the professional team at Chicago Dental Arts for more information on Ultherapy treatment! The ultrasound stimulates collagen production deep beneath the skin’s surface. The body naturally regenerates collagen, and because this process is gradual, you can expect to see results over a period of time. Generally, it takes patients 3-6 months to begin seeing results. Our practitioners are able to see the targeted deeper skin layers while the procedure is taking place, ensuring that your problem areas are addressed.
Our team is happy to answer all of your questions relating to Ultherapy. Call us today for a consultation and no-obligation quote!
Over 16 Years In The Making…
The skin tightening technology behind Ultherapy was created through a research partnership formed in 1998 to develop micro-focused ultrasound for non-invasive treatment of liver cancer. In 2004, Ultherapy was established to develop this technology for aesthetic space. After two years of extensive pre-clinical research, Ultherapy conducted 3 clinical studies to evaluate the safety and efficacy of the technology. One of these studies, an efficacy study at Northwestern University in 2007, demonstrated a clinically significant brow lift in 89% of the patients treated and led to Ultherapy’s first FDA indication. In September 2009, Ultherapy was granted de novo 510K clearance from the FDA, designating the device as a new category of non-invasive technology without a comparable predicate. In 2012, the system received an FDA indication for neck and under-chin lift, and in 2013, it received a third FDA indication for visualization of dermal and subdermal tissue layers.






